 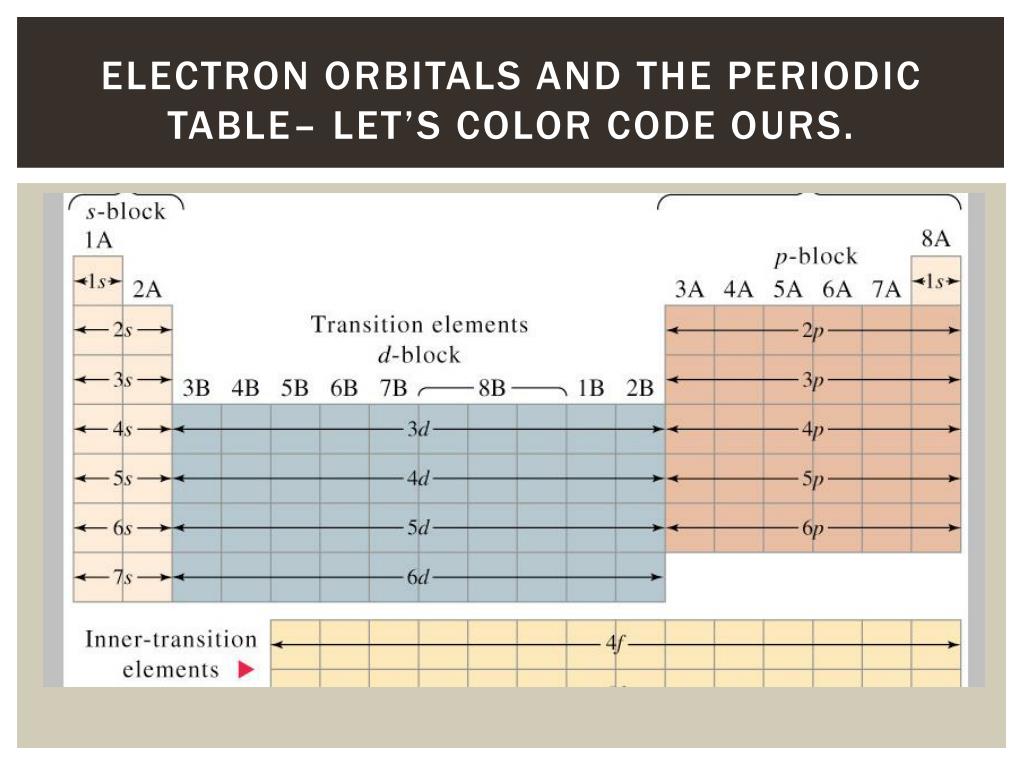
Accordingly, the 4 s orbital is filled prior to the 3 d orbital because of shielding and penetration effects. 
The implicit assumption is that the sum of the atomic orbital energies represents the total energy of the molecule. The periodic table gives the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p3. This 118 element periodic table is a 1920×1080 HD wallpaper. A slightly more complicated example is the electron configuration of bismuth (symbolized Bi, with Z 83). \) provides an alternative method for determining the electron configuration.\) but it is helpful to visualize the atom as a sphere with the nucleus in the center. When you click on any underlined abbreviation for an element, detailed information is displayed in the lower table. In this case, 2+2+6+2+6+2+10+6+2+1 39 and Z39, so the answer is correct. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
